ISO 9001 Traceability Requirements: Complete Implementation Guide
Key Takeaways
- ISO 9001:2015 Section 8.5.2 explicitly requires product identification and traceability
- Traceability means tracking every component from raw material to finished product
- Paper-based traceability systems are audit liabilities — digital systems reduce risk
- Batch-level vs part-level traceability: choose based on your risk profile
- Non-conformance resolution time drops 60-80% with proper digital traceability
- MES systems automate traceability records, eliminating human error
Traceability is one of the most discussed—and most misunderstood—requirements in ISO 9001. Many organizations treat it as a paperwork exercise, generating stacks of records that satisfy auditors but provide little operational value. Others under-invest in traceability only to discover critical gaps during customer complaints, product recalls, or certification audits.
This guide provides a thorough, practical walkthrough of ISO 9001:2015 traceability requirements. We cover what the standard actually demands, how to implement a traceability system that works on the shop floor, how to avoid common pitfalls, and how to prepare for audits with confidence.
What ISO 9001:2015 Says About Traceability
Traceability in ISO 9001:2015 is addressed primarily in Clause 8.5.2 – Identification and Traceability. The clause states:
"The organization shall use suitable means to identify outputs when it is necessary to ensure the conformity of products and services. The organization shall identify the status of outputs with respect to monitoring and measurement requirements throughout production and service provision. The organization shall control the unique identification of the outputs when traceability is a requirement, and shall retain the documented information necessary to enable traceability."
There are three distinct requirements embedded in this clause:
- Identification: You must be able to identify your products and their components at all stages of production.
- Status identification: You must know the inspection and test status of every item—has it been inspected? Did it pass? Is it on hold?
- Traceability: When required (by the organization, by customers, or by regulations), you must maintain unique identification and retain records that enable full traceability.
Note that the standard says "when traceability is a requirement." ISO 9001 does not mandate serialized traceability for every organization. However, it does require that when traceability is needed—whether driven by customer contracts, regulatory requirements, or your own quality system—you must implement and maintain it effectively.
When Traceability Becomes Mandatory
While ISO 9001 leaves traceability partially optional, many industry-specific standards and regulations make it non-negotiable:
Aerospace: AS9100 Rev D
AS9100 significantly expands on ISO 9001's traceability requirements. Clause 8.5.2 in AS9100 requires that organizations "maintain the identification of the configuration of the products and services in order to identify any differences between the actual configuration and the agreed configuration." In practice, this means full serialized traceability with material certifications, process records, and inspection data linked to every individual part or assembly.
Medical Devices: ISO 13485
ISO 13485:2016 Clause 7.5.9 requires that organizations "document procedures for traceability" and that "such procedures shall define the extent of traceability—in accordance with applicable regulatory requirements—and the records required." For implantable devices and Class III products, this extends to tracking every unit from raw material through delivery to the specific patient.
Automotive: IATF 16949
IATF 16949 requires traceability throughout the entire supply chain, with particular emphasis on identification of inspection and test status, control of suspect or non-conforming product, and documented information that enables containment actions when quality issues arise.
Customer-Driven Requirements
Even outside regulated industries, major OEMs and tier-one suppliers increasingly require their vendors to maintain full traceability. Defense contractors, energy companies, and electronics manufacturers commonly include traceability clauses in purchase orders and supplier quality agreements.
What Needs to Be Traced
An effective traceability system must capture data across several dimensions. Here is what you need to track and link together:
Material Traceability
- Raw material certifications (mill certs, certificates of analysis)
- Material lot/batch numbers and heat numbers
- Supplier information and purchase order references
- Incoming inspection results
- Material storage conditions and shelf life (where applicable)
Process Traceability
- Work order and routing information
- Operation sequence and timestamps
- Machine identification and setup parameters
- Operator identification and certification status
- Environmental conditions (temperature, humidity) for sensitive processes
- Tooling and fixture identification
Quality Traceability
- In-process inspection results at each operation
- Final inspection and test data
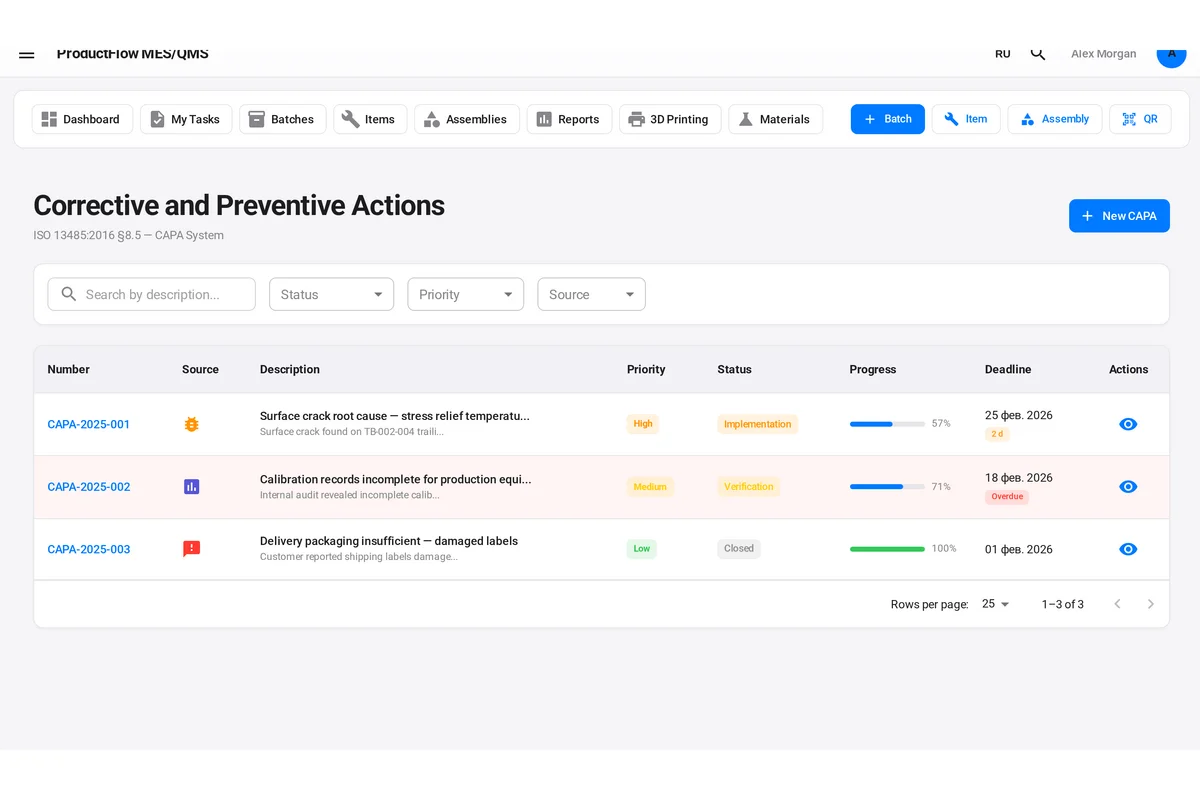
- Non-conformance reports (NCRs) and dispositions
- Corrective action records
- Calibration status of measuring equipment used
Delivery Traceability
- Shipping records linked to serial/lot numbers
- Customer purchase order references
- Certificates of conformance
- Packaging and preservation records
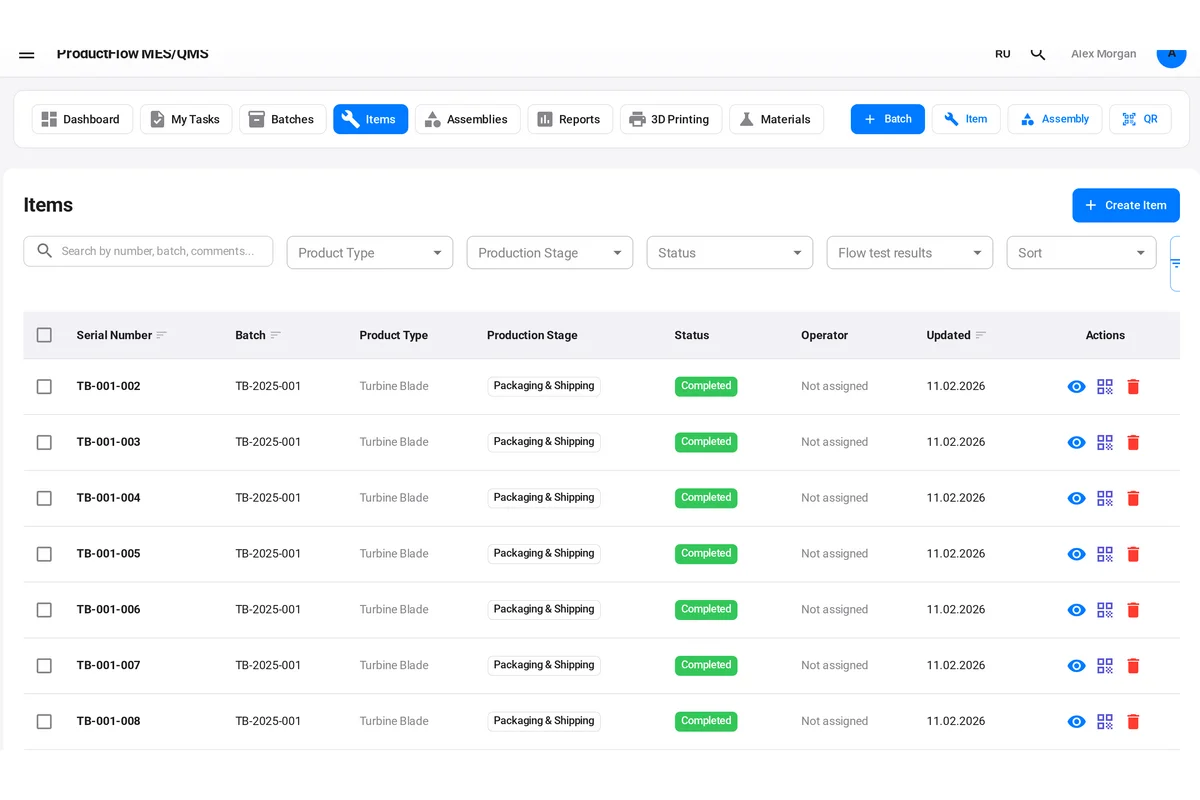
How to Implement Traceability in Manufacturing
Implementing a robust traceability system requires more than buying software. It requires a deliberate approach that integrates people, processes, and technology.
Step 1: Define Your Traceability Scope
Before implementing anything, determine what level of traceability your organization requires. Ask these questions:
- Do customer contracts specify traceability requirements?
- Do applicable regulations require serialized or lot-level traceability?
- What is the risk and cost of a product recall in your industry?
- What traceability level do your internal quality objectives demand?
The answers will tell you whether you need batch/lot-level traceability (grouping products by production run) or serialized traceability (uniquely identifying each individual unit).
Step 2: Design Your Identification System
Every traceable item needs a unique identifier. Common approaches include:
- Serial numbers: Unique alphanumeric codes assigned to individual parts
- Lot/batch numbers: Identifiers assigned to groups of parts produced together
- 2D barcodes or Data Matrix codes: Machine-readable identifiers that encode serial numbers, part numbers, and other data
- RFID tags: For high-value items requiring automated tracking through multiple facilities
Your identification system must be durable enough to survive all production processes. A laser-engraved serial number is useless if it gets machined off in a subsequent operation. Plan your marking location and method carefully.
Step 3: Map Your Data Collection Points
Walk through your production process and identify every point where traceability data must be captured. At each point, define:
- What data is collected (measurements, pass/fail, material consumed, operator ID)
- How data is collected (barcode scan, manual entry, automatic sensor reading)
- Where data is stored (paper traveler, spreadsheet, MES/QMS system)
- Who is responsible for data entry
Step 4: Implement the Technology
While paper-based traceability systems can satisfy ISO 9001 requirements, they are increasingly inadequate for modern manufacturing. Paper systems suffer from illegibility, loss, delayed data entry, and the inability to search or analyze records efficiently.
A digital Manufacturing Execution System (MES) with integrated quality management provides:
- Real-time data capture at the point of production
- Automatic linking of materials, processes, and quality records
- Instant forward and backward traceability queries
- Automated certificate of conformance generation
- Secure, tamper-evident audit trails
Step 5: Train Your Team
The best traceability system in the world fails if operators do not understand why traceability matters and how to use the system correctly. Training should cover:
- Why traceability is critical (use real examples of recalls and quality escapes)
- How to scan, record, and verify traceability data at each workstation
- What to do when something goes wrong (missing label, wrong material, system error)
- How their daily actions directly affect audit readiness and customer satisfaction
Common Traceability Pitfalls
Based on hundreds of quality audits and implementation projects, these are the most frequent traceability failures:
Broken Chain of Custody
Traceability must be continuous from receiving through shipping. A single gap—one operation where the serial number is not recorded, one material transfer without documentation—breaks the entire chain. Auditors specifically look for these gaps by selecting finished products and tracing them backward step by step.
Inconsistent Identification Methods
Using different numbering schemes in different departments or production areas creates confusion and errors. Establish a single, organization-wide identification standard and enforce it everywhere.
Relying on Memory or Tribal Knowledge
When operators say "We always use material from the same bin, so we know it is the right lot," you have a traceability gap. Every material usage must be positively verified and recorded, regardless of how routine the operation is.
Retroactive Record Creation
Creating traceability records after the fact ("backfilling") is a serious quality system failure. Records must be created at the time the activity occurs. Auditors can detect retroactive entries by examining timestamps, handwriting patterns, and data entry sequences.
Inadequate Retention
ISO 9001 requires that documented information be retained, but it does not specify a retention period—your organization must define this based on product lifecycle, warranty periods, contractual obligations, and regulatory requirements. Aerospace and medical device records are commonly retained for 10 to 30 years or more.
Preparing for Traceability Audits
Auditors assess traceability by performing trace exercises. They will typically:
- Select a finished product from your shipping records or warehouse.
- Request a backward trace: "Show me every material, process step, inspection, and operator involved in producing this specific unit."
- Request a forward trace: "This raw material lot was received on January 15. Show me every product that used material from this lot and where those products were shipped."
- Check status identification: "Show me which items in your WIP area have been inspected and which are awaiting inspection."
- Verify links: Confirm that material certifications match the lot numbers recorded against finished products, that operator signatures match authorized personnel lists, and that calibration records for inspection equipment are current.
To prepare effectively, conduct internal trace exercises regularly—at least quarterly. Time yourself: a complete backward trace should take minutes, not hours. If it takes longer, your system has gaps that need addressing before the registrar arrives.
Documentation Tips for Audit Success
- Maintain a traceability procedure that clearly defines your identification methods, data collection points, and retention requirements.
- Keep records organized and accessible—whether digital or physical, every record must be retrievable within a reasonable time.
- Document your trace exercise results as evidence of system effectiveness during management reviews.
- Ensure non-conforming product is physically segregated and clearly identified (quarantine areas with distinct marking).
Traceability Beyond Compliance: The Business Value
While regulatory compliance drives most traceability implementations, the business benefits extend far beyond passing audits:
Reduced recall scope: With precise traceability, you can identify exactly which units are affected by a quality issue. Instead of recalling an entire year's production, you recall only the specific lot or serial number range. This can reduce recall costs by 90% or more.
Faster root cause analysis: When a customer complaint arrives, traceability records let you quickly identify the material source, process conditions, and personnel involved. What previously took weeks of investigation now takes hours.
Supply chain accountability: Traceability data holds suppliers accountable for material quality. When you can prove that a defect originated from a specific material lot from a specific supplier, you have the data to support corrective action requests and, if necessary, supplier changes.
Customer confidence: Providing detailed certificates of conformance with full traceability data differentiates your organization from competitors who rely on generic batch records. It builds trust and supports premium pricing.
Achieve Full Traceability with ProductFlow
ProductFlow provides serialized traceability, real-time quality tracking, and automated certificate generation—everything you need for ISO 9001, AS9100, and ISO 13485 compliance. See pricing or explore ProductFlow features.
Key Takeaways
- ISO 9001:2015 Clause 8.5.2 requires identification, status identification, and traceability (when required by your context).
- Industry standards like AS9100, ISO 13485, and IATF 16949 make detailed traceability mandatory, not optional.
- Effective traceability links materials, processes, quality records, and delivery data into a continuous, auditable chain.
- Digital systems (MES/QMS) dramatically outperform paper-based approaches in reliability, speed, and audit readiness.
- Regular internal trace exercises are the single best way to prepare for certification audits and identify system gaps.
- The business value of traceability—reduced recalls, faster root cause analysis, supply chain accountability—far exceeds the compliance cost.
Traceability is not just a checkbox on an audit form. Implemented well, it becomes a strategic asset that protects your customers, your reputation, and your bottom line. The key is to start with a clear scope, invest in the right tools, train your people, and verify your system continuously through internal trace exercises.